Generation of TXXY and PXXY Diagrams for Immiscible Systems Using Aspen Plus Binary Analysis
Project Description
Phase behavior analysis of partially or completely immiscible systems is essential in separation process design, particularly for liquid-liquid extraction and heterogeneous azeotropic distillation. This project demonstrates how to generate TXXY (Temperature–Composition) and PXXY (Pressure–Composition) plots for immiscible binary systems using the enhanced Binary Analysis tools available in Aspen Plus (Version 11 and higher).
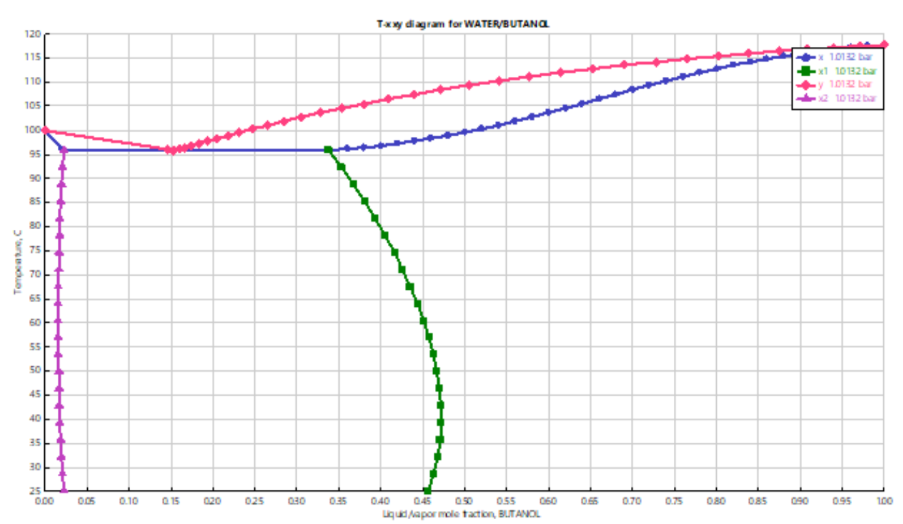
Traditional Txy or Pxy diagrams represent vapor-liquid equilibrium (VLE) only. However, immiscible or partially miscible systems require simultaneous representation of vapor-liquid and liquid-liquid equilibrium (LLE). The TXXY and PXXY analysis types extend conventional diagrams by including two-liquid phase compositions (x₁ and x₂) along with vapor composition (y), providing a complete thermodynamic picture of two-liquid systems at fixed pressure or temperature.
Using a representative butanol–water system as an example, this project illustrates how to define lower temperature limits for LLE calculations, adjust temperature intervals to accurately capture the upper consolute temperature, and avoid solid-phase regions by constraining the temperature range appropriately.
Process Flow Diagarm
Optimization Strategy
Accurate generation of TXXY diagrams requires careful selection of temperature bounds and interval resolution. The lower temperature limit must be specified to enable liquid-liquid equilibrium calculations, while the upper limit should be chosen to fully capture the two-liquid region without extending into solid-phase zones.
Increasing the number of temperature intervals improves resolution near the upper consolute temperature (critical solution temperature), where the two liquid phases merge into one. Sensitivity analysis on temperature step size ensures accurate identification of phase boundaries and inflection points, which are crucial for separation design and solvent selection.
Binary Analysis Configuration
Aspen Plus Version 11 and above includes specialized binary analysis types:
Txx
Txxy
Pxxy
These allow simultaneous VLE and LLE calculations for immiscible or partially miscible systems.
TXXY Diagram Fundamentals
TXXY represents:
Temperature (T)
Vapor composition (y)
Liquid phase compositions (x₁ and x₂)
The plot shows both vapor-liquid and liquid-liquid equilibrium regions at constant pressure.
Handling Immiscibility and Consolute Points
Proper selection of temperature limits ensures:
Accurate detection of two-liquid regions
Identification of upper consolute temperature
Avoidance of solid-phase transition zones
Projects Insight
Importance of LLE Modeling
Essential for immiscible systems
Supports liquid-liquid extraction design
Captures phase splitting behavior
Enhanced Binary Tools (V11+)
Txx includes LLE only
Txxy includes VLE + LLE
Pxxy operates at constant temperature
Upper Consolute Temperature Detection
Requires fine temperature discretization
Indicates merging of two liquid phases
Critical for solvent selection
Solid-Phase Region Awareness
Inflection points may indicate solid formation
Lower temperature bound prevents instability
Improves diagram clarity
Application to Butanol–Water System
Classic partially miscible system
Exhibits two-liquid region
Demonstrates combined VLE–LLE behavior
Industrial Applications
Heterogeneous azeotropic distillation
Decanter design
Extraction solvent evaluation
Conclusion
The aerobic batch fermentation process for 1,4-Butanediol production integrates controlled microbial growth, regulated substrate feeding, and optimized oxygen mass transfer to achieve high product yield and operational stability. By enhancing kLa and aligning substrate supply with metabolic demand, the process improves conversion efficiency while minimizing impurityformation. Implementation of structured control strategies ensures reproducible batch performance and industrial scalability. Collectively, the optimized framework strengthens the feasibility of renewable BDO production and supports sustainable chemical manufacturing advancement.
