Aspen Plus Simulation of Methanol Synthesis Process
Project Description
Methanol is a critical industrial chemical, widely utilized as a fuel, solvent, and intermediate for producing formaldehyde, acetic acid, and other petrochemicals. This project simulates the steady-state methanol synthesis process using Aspen Plus, transforming syngas—a mixture primarily of hydrogen, carbon monoxide, and carbon dioxide—into high-purity methanol under high-pressure and moderate-temperature conditions over a copper-based catalyst. The simulation captures the behavior of a real industrial plant, including feed compression, heat exchange, catalytic reactor operation, product cooling, gas–liquid separation, recycle streams, and distillation purification.
The core of the process lies in the catalytic reactor, where the copper-based catalyst facilitates the exothermic conversion of CO and CO₂ into methanol. Industrial reactor configurations such as multi-bed quench reactors and two-stage reactor systems are modeled to analyze the effects of temperature control, conversion efficiency, and heat management. Heat integration between reactor effluent and incoming feed streams is included to improve energy efficiency and reduce external utility consumption.
The objective of this simulation is to evaluate process performance, optimize operating conditions, and provide accurate mass and energy balances. By replicating real industrial operations, the model supports design studies and performance analysis, enabling assessment of reaction behavior, operational efficiency, and overall methanol yield under different process conditions.
Process Flow Diagarm
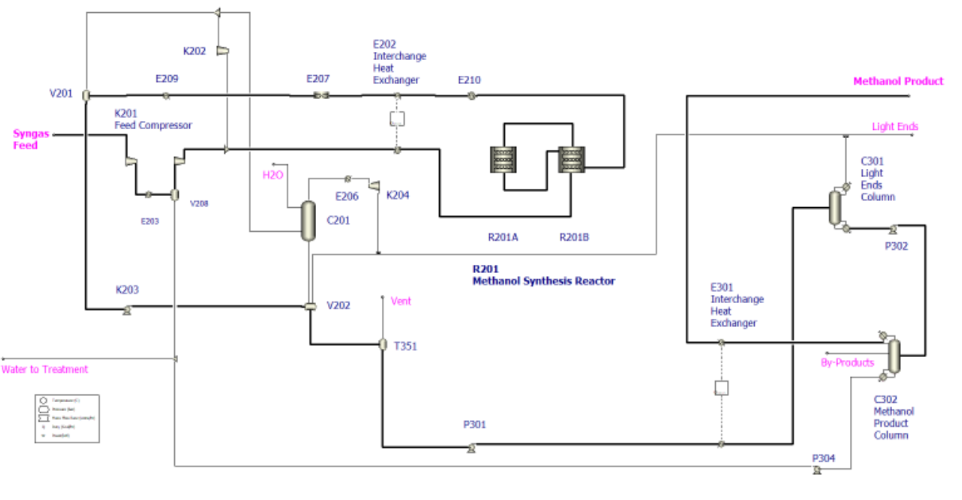
Optimization Strategy
The operational strategy focuses on maximizing methanol production while ensuring stable reactor performance and extending catalyst life. Reactor temperature is carefully controlled through quench gas injection or heat removal systems to prevent hotspots and avoid catalyst deactivation. Simultaneously, feed composition is monitored to maintain the optimal hydrogen-to-carbon ratio, which ensures efficient reaction kinetics and minimizes the need for excessive recycle streams. Pressure control is applied throughout the system to achieve high conversion rates, while purge streams are used to prevent the accumulation of inert gases that could lower process efficiency
In addition, heat integration plays a critical role in improving the overall energy efficiency of the plant. By exchanging heat between reactor effluent and incoming feed streams, the process reduces external utility demand and maintains consistent thermal management. This approach not only optimizes energy consumption but also stabilizes industrial-scale operations, ensuring reliable methanol production under varying feed and operating conditions.
Process Components and Feed System
The electrolyte system is modeled using the Electrolyte Non-Random Two-Liquid (ElectrolyteNRTL) property method, which accurately represents chemical reactions and phase behavior inaqueous amine solutions. Vapor-phase properties are estimated using a suitable equation ofstate for gas mixtures. The absorber and stripper are simulated using a rate-based approachthat accounts for mass transfer, heat transfer, and reaction kinetics, providing more realisticpredictions than equilibrium-based models
Column Design and Hydraulic Performance
The absorber consists of multiple packed sections to maximize gas–liquid contact, while thestripper utilizes valve trays for efficient solvent regeneration. Hydraulic analysis evaluatesflooding limits, pressure drop, and stage or packing efficiency. This assessment helps identifyoperational constraints and ensures stable column performance under actual plant conditions.
Process Performance and Validation Results
Model results are compared with plant data for key parameters including solvent loading, temperature distribution, CO₂ capture rate, steam consumption, and off-gas composition. The simulation shows strong agreement with operating data and confirms a capture capacity of approximately 130 tpd of CO₂. Economic evaluation estimates operating and capital costs, while carbon tracking assesses overall emission reduction performance and potential carbon costbenefits.
Projects Insight
Accuracy of Rate-Based Modeling
- Rate-based columns provide better prediction than equilibrium models.
- They capture mass transfer and reaction limitations effectively.
- Essential for realistic design and plant validation.
Hydraulic Constraints Importance
- Column flooding or high pressure drop limits capacity.
- Hydraulic analysis ensures safe and stable operation.
- Design adjustments may be required for debottlenecking
Energy Consumption in Solvent Regeneration
- Steam demand in the stripper is the major operating cost.
- Heat integration significantly reduces energy requirement.
- Optimization of reboiler duty improves process economics.
Carbon Reduction Performance
- The process captures a significant portion of flue gas CO₂.
- Net emissions can become negative with proper operation.
- Supports environmental compliance and carbon management.
Solvent Loading Impact
- Lean and rich loading determine absorption capacity.
- Improper loading increases circulation rate and energy use.
- Optimal loading improves overall efficiency
Solvent Degradation Considerations
- MEA degradation occurs due to heat and impurities.
- Makeup solvent is required to maintain performance.
- Proper temperature and impurity control extend solvent life.
Conclusion
The Aspen Plus simulation for MEA-based CO₂ capture successfully validates plant performance and provides a reliable representation of industrial operating conditions. By integrating accurate electrolyte thermodynamics, rate-based column modeling, and hydraulic evaluation, the study captures the complex interactions governing absorption and solvent regeneration. Implementation of a balanced operational strategy improves CO₂ removal efficiency while reducing steam consumption and overall operating cost. The validated model serves as a robust platform for process optimization, capacity enhancement, and energy integration studies. Ultimately, this work supports the development of efficient and economically viable carbon capture systems, contributing to sustainable emission reduction and improved environmental performance in large-scale industrial applications.
