Estimation of the Eutectic Point of Binary Mixtures Using Aspen Plus
Project Description
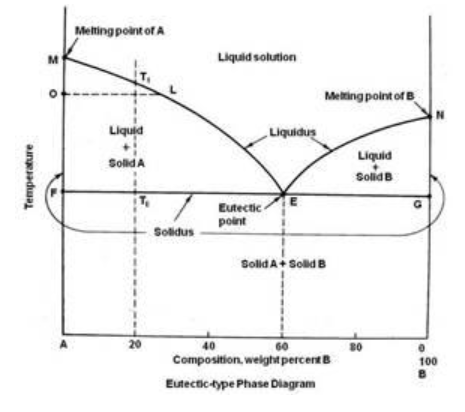
The eutectic point of a mixture represents the specific composition and temperature at which a mixture of substances melts or solidifies simultaneously at the lowest possible temperature. Understanding the eutectic behavior of chemical mixtures is essential in many industrial processes such as metallurgy, energy storage systems, and chemical manufacturing. This project focuses on estimating the eutectic point of a binary mixture using Aspen Plus simulation tools.
In this study, the NaNO₃–KNO₃ system, commonly referred to as solar salt, is modeled to analyze the melting and solidification behavior of the mixture. Aspen Plus uses a Gibbs reactor to simulate the thermodynamic equilibrium between the solid and liquid phases of the mixture. By defining the components as both conventional and solid components, the simulation allows equilibrium calculations that accurately represent the physical and chemical behavior of the system.
The simulation approach involves performing a sensitivity analysis by gradually reducing the reactor temperature while monitoring the liquid and solid phase fractions. When the liquid fraction decreases and the solid fraction begins to form simultaneously, the eutectic point is reached. This method enables engineers to predict eutectic temperatures and compositions efficiently without extensive experimental measurements
Process Flow Diagarm
Optimization Strategy
The operational strategy of this simulation involves modeling the solid–liquid equilibrium of the NaNO₃–KNO₃ mixture using a Gibbs reactor in Aspen Plus. The reactor is initially set at a temperature above the melting points of both components to ensure that the mixture begins in a completely molten state.
This condition provides a stable starting point for analyzing the transition from liquid to solid phases as the temperature is gradually decreased. A sensitivity analysis is then applied to vary the reactor temperature over a specified range while monitoring key variables such as liquid fraction, solid fraction, and component mass fractions. The eutectic point is identified when the solid phase begins to appear while the liquid phase simultaneously decreases. This method allows accurate estimation of the eutectic temperature and helps evaluate the phase behavior of the binary mixture.
Simulation of Solid–Liquid Equilibrium for Solar Salt Mixtures Using Aspen Plus
This project investigates the phase behavior of solar salt mixtures consisting of sodium nitrate and potassium nitrate using Aspen Plus simulation software. By modeling the solid–liquid equilibrium in a Gibbs reactor, the study predicts the melting and solidification behavior of the mixture and identifies the eutectic temperature. The simulation results provide valuable insights for applications such as thermal energy storage and industrial heat transfer systems.
Thermodynamic Analysis of Binary Salt Mixtures for Eutectic Point Prediction
This project focuses on thermodynamic modeling of binary salt mixtures to determine their eutectic composition and temperature using Aspen Plus. Through equilibrium calculations and sensitivity analysis, the model identifies the conditions at which both components solidify simultaneously. The results help engineers understand phase transitions and improve the design of high-temperature energy storage materials.
Modeling Phase Equilibrium of Molten Salt Systems Using Aspen Plus
This project demonstrates the application of Aspen Plus for modeling phase equilibrium in molten salt systems. By simulating the melting and solidification processes of nitrate salt mixtures, the model estimates eutectic behavior and evaluates phase transitions across a temperature range. The study supports the development of efficient energy storage systems and advanced thermal management technologies.
Projects Insight
Understanding the Eutectic Point
- Represents the lowest melting temperature of a mixture
- Occurs at a specific composition of components
- Important for predicting phase transition behavior
Sensitivity Analysis for Phase Study
- Allows systematic variation of reactor temperature
- Tracks liquid and solid fraction changes
- Identifies temperature where solidification begins
Role of Gibbs Reactor in Aspen Plus
- Performs equilibrium calculations for multiple phases
- Handles solid–liquid equilibrium modeling
- Determines phase composition at different temperatures
Industrial Applications of Eutectic Systems
- Thermal energy storage in solar power plants
- Metallurgical alloy production
- Chemical and materials engineering processes
Importance of Solid Property Methods
- Calculates thermodynamic properties of solid mixtures
- Uses equations such as the Barin equation
- Ensures accurate prediction of enthalpy and phase behavior
Advantages of Simulation-Based Prediction
- Reduces need for expensive laboratory experiments
- Enables quick analysis of multiple compositions
- Improves process design and optimization
Conclusion
The estimation of eutectic points in binary mixtures is an essential aspect of thermodynamic analysis in chemical and materials engineering. This project demonstrates how Aspen Plus can be effectively used to simulate the melting and solidification behavior of mixtures such as NaNO₃ and KNO₃ using a Gibbs reactor model. By performing sensitivity analysis and monitoring phase fractions during temperature variation, the eutectic temperature and composition of the mixture can be identified with reasonable accuracy. The simulation approach offers a practical and efficient alternative to experimental methods, allowing engineers to study complex phase behavior and optimize industrial processes. Understanding eutectic properties is particularly valuable in applications such as thermal energy storage systems, metallurgical operations, and advanced material development, where accurate knowledge of melting and solidification characteristics plays a critical role in improving system performance and efficiency.
