Integrated Cumene Production Process with Reaction Kinetics, Separation, Recycle, and Plantwide Economic Optimization
Project Description
Cumene (isopropylbenzene) is industrially produced by the alkylation of benzene with propylene. In this process, benzene reacts with propylene to form cumene, while a secondary reaction between cumene and propylene produces an undesirable byproduct, p-diisopropyl benzene (PDIB). This project presents a rigorous Aspen Plus model incorporating reaction kinetics, separation units, recycle streams, and purification systems to study the economic and operational trade-offs in cumene production.
The kinetic reactor model captures the competing reactions, where the secondary PDIB formation has a higher activation energy than the primary cumene reaction. Lower reactor temperatures favor higher cumene selectivity but require larger reactor volumes. The model
includes propane impurity in the propylene feed, which is inert and removed through a flash drum (V-701). The flashed gas stream, containing propane and unreacted propylene, is purged and utilized as fuel, making propylene conversion economically significant.
Downstream purification includes two distillation columns. Column T-601 recovers unreacted benzene as distillate for recycle to the reactor feed, while Column T-602 separates high-purity cumene as the overhead product. PDIB is removed in the bottoms stream and may be used for fuel recovery. A design specification is implemented to control benzene feed flow to achieve a targeted conversion or production rate. The model provides a complete framework for plantwide economic evaluation and optimization studies.
Process Flow Diagarm
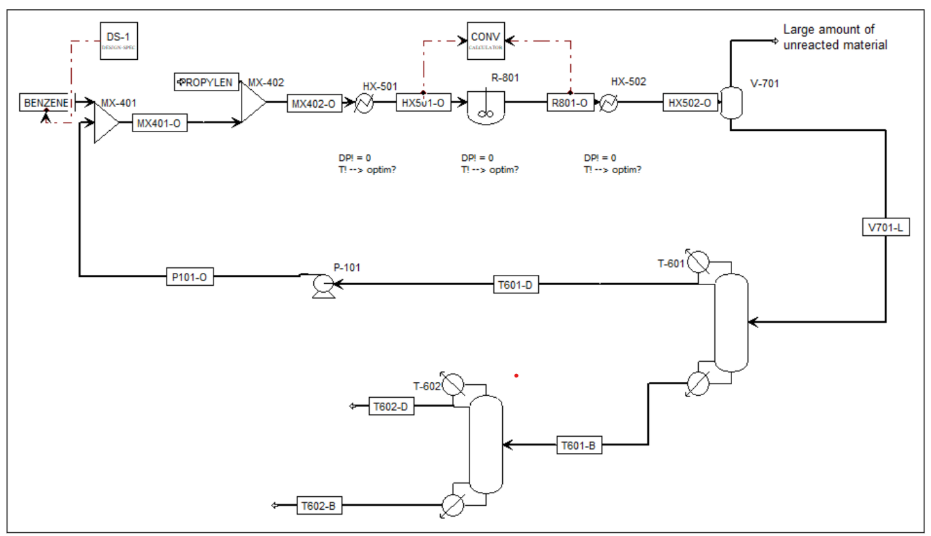
Optimization Strategy
The optimization strategy focuses on balancing reactor operating temperature, reactor size, selectivity, and recycle flow rate to minimize total annual cost. Since PDIB formation increases at higher temperatures, reactor temperature optimization directly affects selectivity and downstream separation load. Lower temperatures improve selectivity but increase capital investment due to larger reactor volume requirements.
Recycle optimization plays a crucial role in plantwide economics. Higher recycle ratios improve reactant utilization but increase energy consumption in separation units. Flash and distillation systems must be optimized to minimize energy demand while maintaining high purity cumene production. By integrating material balances, energy balances, and kinetic constraints, the model supports comprehensive economic trade-off analysis
Reaction Kinetics and Selectivity Control
The reactor model incorporates temperature-dependent kinetic expressions for both primary
and secondary reactions. Since PDIB formation has a higher activation energy, temperature control becomes a key lever for improving selectivity toward cumene.
Separation and Purification System Design
Two distillation columns recover unreacted benzene and purify cumene product. Efficient separation reduces raw material consumption and ensures high product quality, while managing
energy requirements.
Plantwide Recycle and Economic Trade-Offs
Recycle of benzene enhances raw material efficiency but increases column duties and operating cost. Economic optimization balances conversion, recycle ratio, and equipment sizing.
Projects Insight
Competing Reaction Dynamics
● Primary: Benzene + Propylene → Cumene
● Secondary: Cumene + Propylene → PDIB
● Higher activation energy for PDIB formation
Temperature–Selectivity Trade-Off
● Lower temperature increases selectivity
● Larger reactor volume required
● Impacts capital investment
Propylene Conversion Importance
● Propane impurity exits via flash drum
● Unreacted propylene burned as fuel
● High conversion improves profitability
Benzene Recycle Strategy
● T-601 recovers benzene overhead
● Reduces fresh feed requirement
● Controlled via design specification
Cumene Purification
● T-602 produces high-purity cumene
● PDIB removed as bottoms product
● Energy-intensive separation
Plantwide Economic Sensitivity
● Reactor size vs. energy demand
● Recycle rate vs. column duty
● Selectivity vs. overall cost
Conclusion
This project presents a rigorous kinetic and separation-based model of the cumene production process, integrating reactor design, purification, recycle, and economic evaluation. The interaction between reaction kinetics and separation design introduces classical engineering trade-offs involving temperature, selectivity, reactor size, and recycle flow rate. By incorporating design specifications and plantwide balances, the model enables systematic optimization of capital and operating costs. The developed framework provides a comprehensive educational and industrial reference for simultaneous process design, control strategy development, and economic analysis in aromatic alkylation systems.
