Validation of Plant Data for CO₂ Capture from Flue Gas Using MEA
Project Description
Carbon capture through chemical absorption is one of the most established technologies for reducing greenhouse gas emissions from large industrial sources. This project focuses on the validation of plant operating data for post-combustion CO₂ capture from flue gas using monoethanolamine (MEA) as the solvent. The system is designed to remove approximately 130 tonnes per day of CO₂ from a coal-fired power plant exhaust stream, supporting emission reduction and environmental compliance objectives.
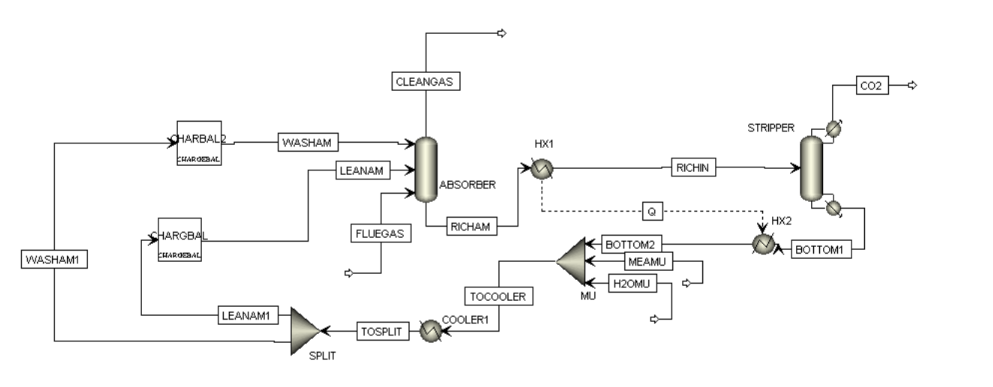
In this process, flue gas containing CO₂, nitrogen, and oxygen enters an absorber column where it flows counter-currently to a lean MEA solvent. CO₂ is selectively absorbed through chemical reaction, producing a treated gas stream with reduced carbon content. The CO₂-rich solvent exiting the absorber is preheated and directed to a stripper column, where thermal regeneration releases high-purity CO₂ and restores the solvent for reuse. The regenerated lean solvent is cooled and recycled back to the absorber, forming a continuous capture loop.
The simulation is developed in Aspen Plus to replicate plant operating conditions and evaluate system performance under real industrial constraints. Model predictions are compared with actual plant measurements, including solvent loading, temperature profiles, steam consumption, and CO₂ removal efficiency. This validation framework ensures reliable process representation and provides a strong basis for performance analysis, optimization, and future process improvement
Process Flow Diagarm
Optimization Strategy
The operational strategy focuses on maximizin The optimization strategy aims to maintain high CO₂ capture efficiency while minimizing energy consumption and ensuring stable column operation. Solvent circulation rate and lean solvent loading are carefully adjusted to provide sufficient absorption capacity without increasing regeneration energy demand. Optimizing the balance between absorption performance and reboiler duty directly improves overall process efficiency and operating cost.
Additional improvements focus on thermal and hydraulic stability within the system. Steam supply to the stripper is regulated to achieve effective solvent regeneration while preventing thermal degradation of MEA. Heat integration between rich and lean solvent streams reduces external energy requirements and enhances thermal efficiency. At the same time, column hydraulic limits such as flooding tendency and pressure drop are monitored to ensure reliablelong-term operation. Solvent makeup and impurity control are also incorporated to compensate for degradation losses and maintain consistent plant performance g methanol production while ensuring stable reactor performance and extending catalyst life. Reactor temperature is carefully controlled through quench gas injection or heat removal systems to prevent hotspots and avoid catalyst deactivation. Simultaneously, feed composition is monitored to maintain the optimal hydrogen-to-carbon ratio, which ensures efficient reaction kinetics and minimizes the need for excessive recycle streams. Pressure control is applied throughout the system to achieve high conversion rates, while purge streams are used to prevent the accumulation of inert gases that could lower process efficiency.
Thermodynamic and Model Framework
The methanol synthesis process utilizes syngas containing hydrogen (H₂), carbon monoxide (CO), carbon dioxide (CO₂), methane (CH₄), water (H₂O), and inert gases. Methanol (CH₃OH) is the primary product, while minor by-products such as ethanol and dimethyl ether (DME) are also formed. The feed gas is compressed to high pressures, typically 50–90 bar, and preheated using heat exchangers with reactor effluent. Recycle gas streams are combined with fresh feed to optimize conversion and maintain the hydrogen-to-carbon ratio essential for efficient reaction kinetics.
Thermodynamic and Physical Property Modeling
The Soave–Redlich–Kwong (SRK) equation of state is used for calculating phase behavior and thermodynamic properties, as it is well-suited for high-pressure gas systems. Binary interaction parameters are tuned using experimental vapor–liquid equilibrium data for the methanol–water system. Accurate thermodynamic modeling is critical for predicting equilibrium conversion, heat effects, and separation performance. Proper property selection enhances reactor temperature prediction and distillation column operation.
Reaction Kinetics and Reactor Modeling
Methanol synthesis primarily occurs through the following reactions:
● CO₂ + 3H₂ ⇌ CH₃OH + H₂O
● CO₂ + H₂ ⇌ CO + H₂O (Reverse Water Gas Shift)
Side reactions forming ethanol and DME are also considered. The main reactions are modeled
using Langmuir–Hinshelwood–Hougen–Watson (LHHW) kinetics to describe heterogeneous
catalytic behavior. Packed-bed reactors are represented using plug flow models, where catalyst
weight, residence time, and operational parameters are defined to replicate industrial
performance accurately.
Industrial Process Configuration
Two industrial reactor configurations are evaluated: ICI Synetix Quench Reactor System
● Multi-bed reactor with interstage cold gas injection to control temperature rise.
● Maintains catalyst activity and improves conversion efficiency.
Lurgi Two-Stage Reactor System
● First stage operates near-isothermally with boiling water cooling.
● Second stage uses gas cooling for additional temperature management.
● Enhances overall conversion and enables efficient heat recovery for steam generation. Downstream operations include product cooling, gas–liquid separation, recycle management,
purge control, and methanol purification through distillation columns.
Projects Insight
Accuracy of Rate-Based Modeling
● Rate-based columns provide better prediction than equilibrium models.
● They capture mass transfer and reaction limitations effectively.
● Essential for realistic design and plant validation.
Hydraulic Constraints Importance
● Column flooding or high pressure drop limits capacity.
● Hydraulic analysis ensures safe and stable operation.
● Design adjustments may be required for debottlenecking.
Energy Consumption in Solvent Regeneration
● Steam demand in the stripper is the major operating cost.
● Heat integration significantly reduces energy requirement.
● Optimization of reboiler duty improves process economics.
Carbon Reduction Performance
● The process captures a significant portion of flue gas CO₂.
● Net emissions can become negative with proper operation.
● Supports environmental compliance and carbon management.
Solvent Loading Impact
● Lean and rich loading determine absorption capacity.
● Improper loading increases circulation rate and energy use.
● Optimal loading improves overall efficiency.
Solvent Degradation Considerations
● MEA degradation occurs due to heat and impurities.
● Makeup solvent is required to maintain performance.
● Proper temperature and impurity control extend solvent life.
Conclusion
The Aspen Plus simulation for MEA-based CO₂ capture successfully validates plant performance and provides a reliable representation of industrial operating conditions. By integrating accurate electrolyte thermodynamics, rate-based column modeling, and hydraulic
evaluation, the study captures the complex interactions governing absorption and solvent regeneration. Implementation of a balanced operational strategy improves CO₂ removal efficiency while reducing steam consumption and overall operating cost. The validated model serves as a robust platform for process optimization, capacity enhancement, and energy integration studies. Ultimately, this work supports the development of efficient and economically viable carbon capture systems, contributing to sustainable emission reduction and improved environmental performance in large-scale industrial applications.
